Design Controls: Building Objective Evidence and Process Architecture to Meet FDA and ISO Compliance - OMTEC 2018 | PPT

9 Tips for Addressing the Documentation Burden of the FDA's Design Control Regulation - Medical Design Briefs

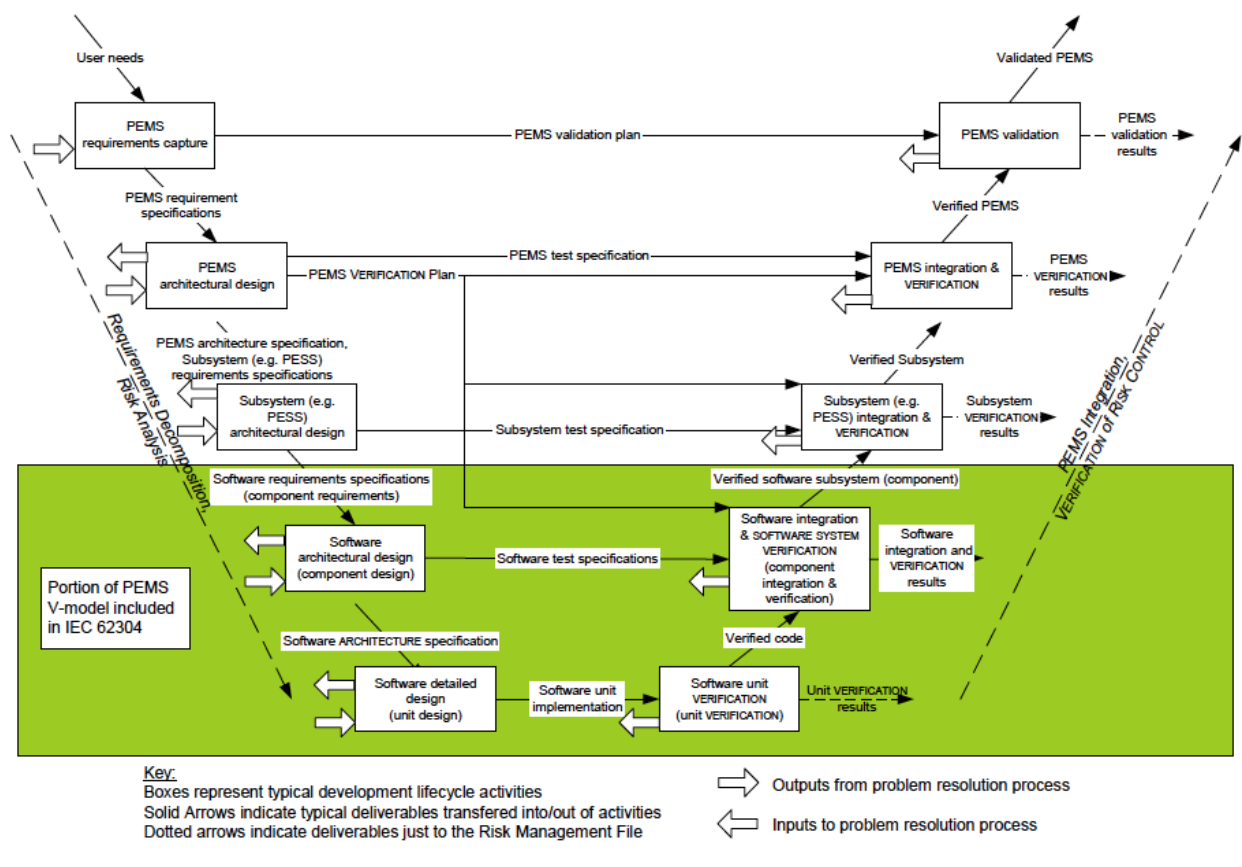
Agile Development in Regulated Environments Example: Medical Devices – Waterfall Lifecyle Model | Scaling Software Agility
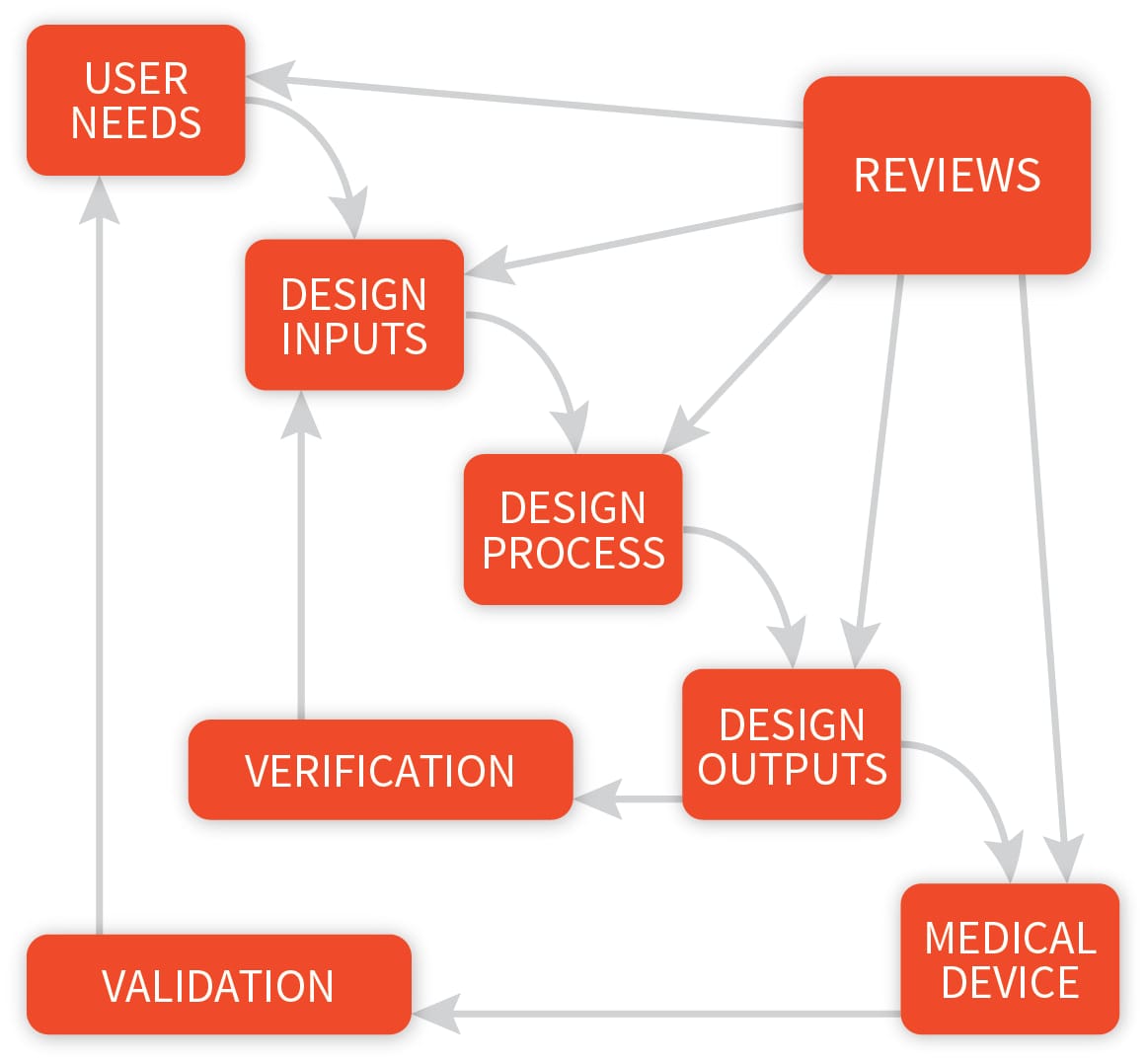
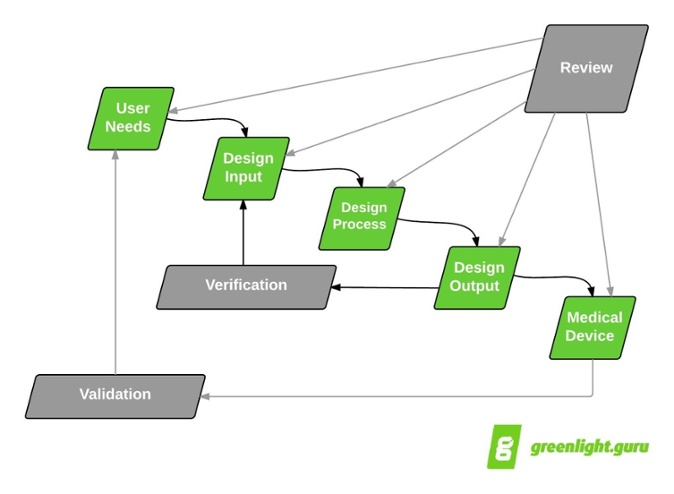
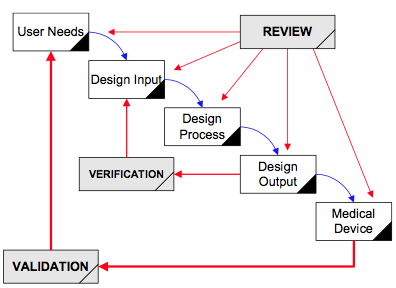
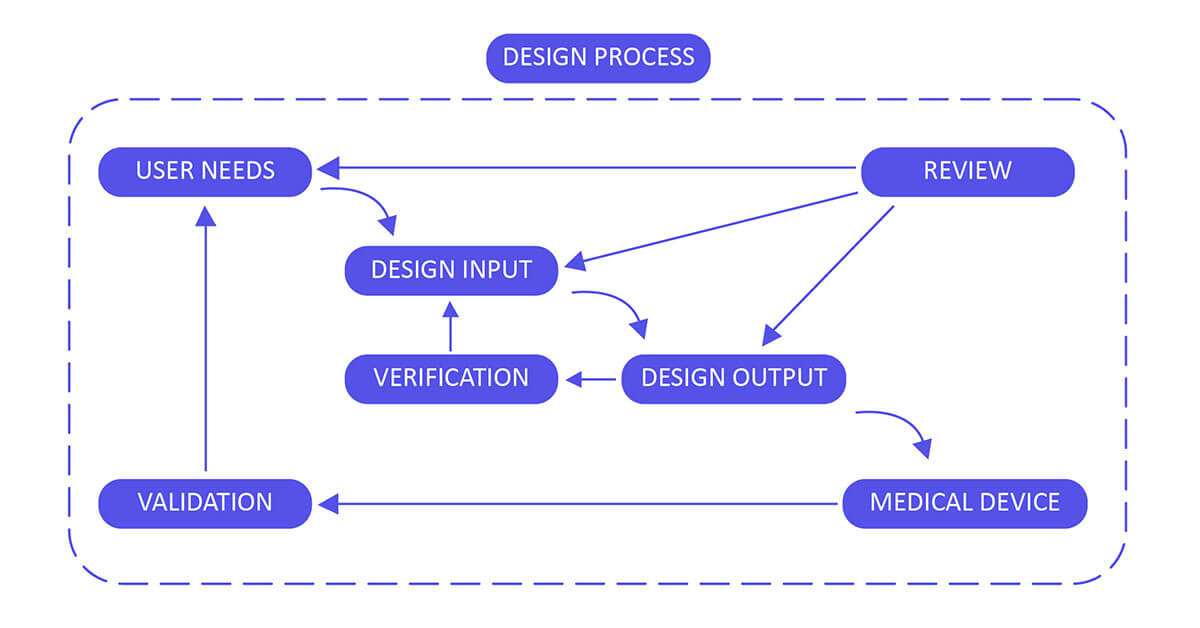
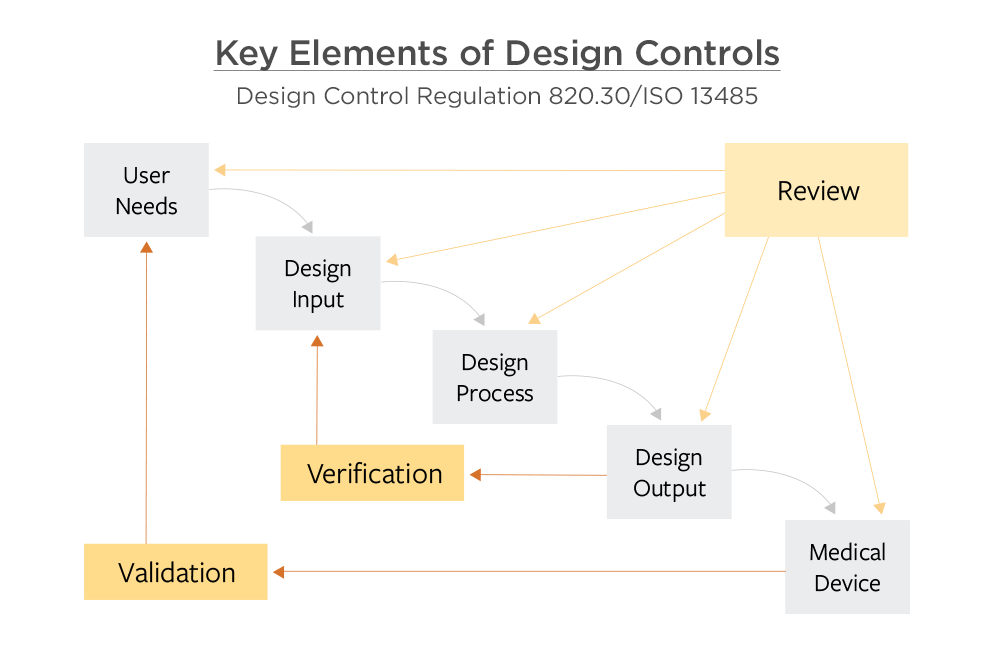
Application of design controls to design process. Design review should... | Download Scientific Diagram

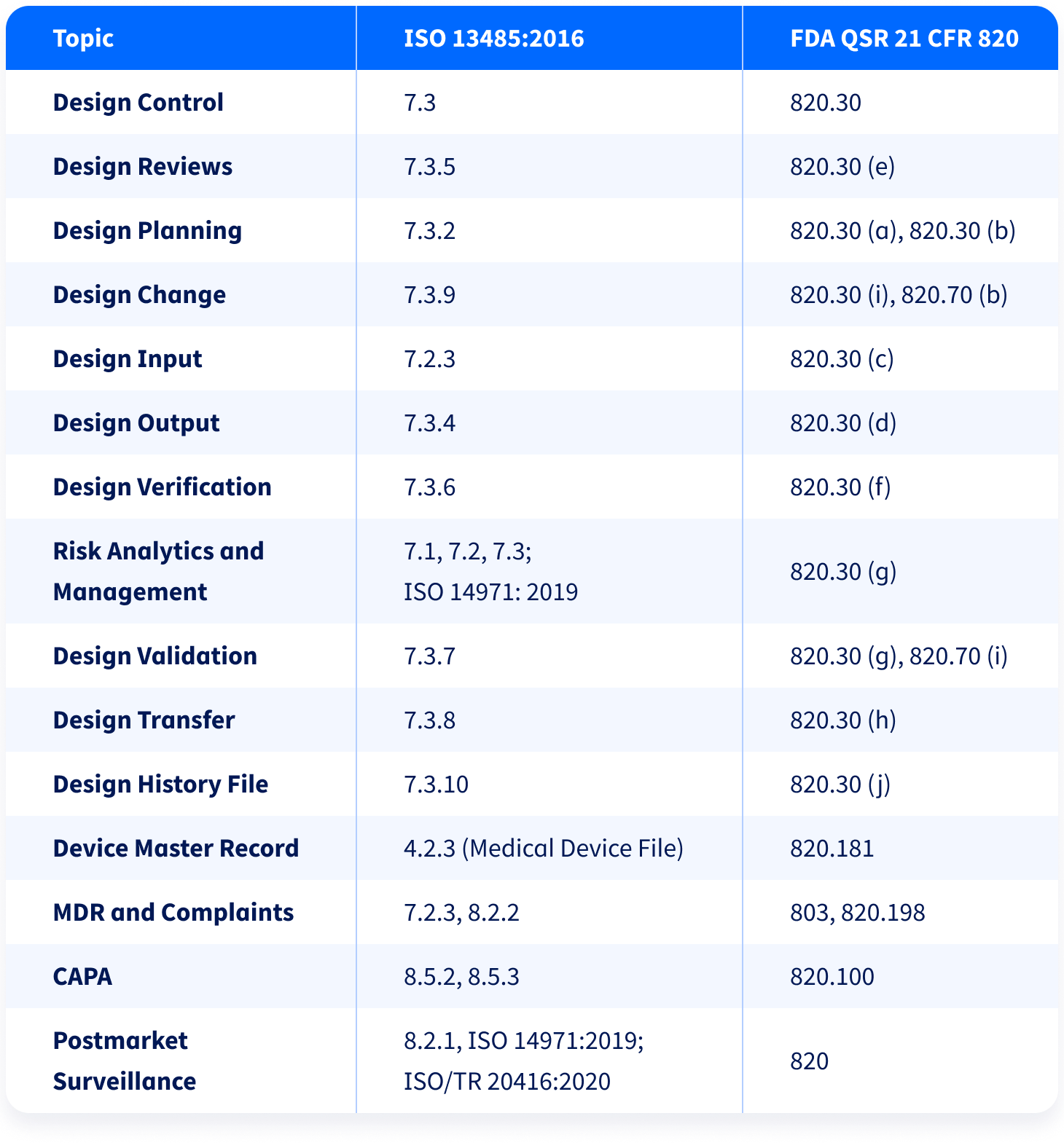
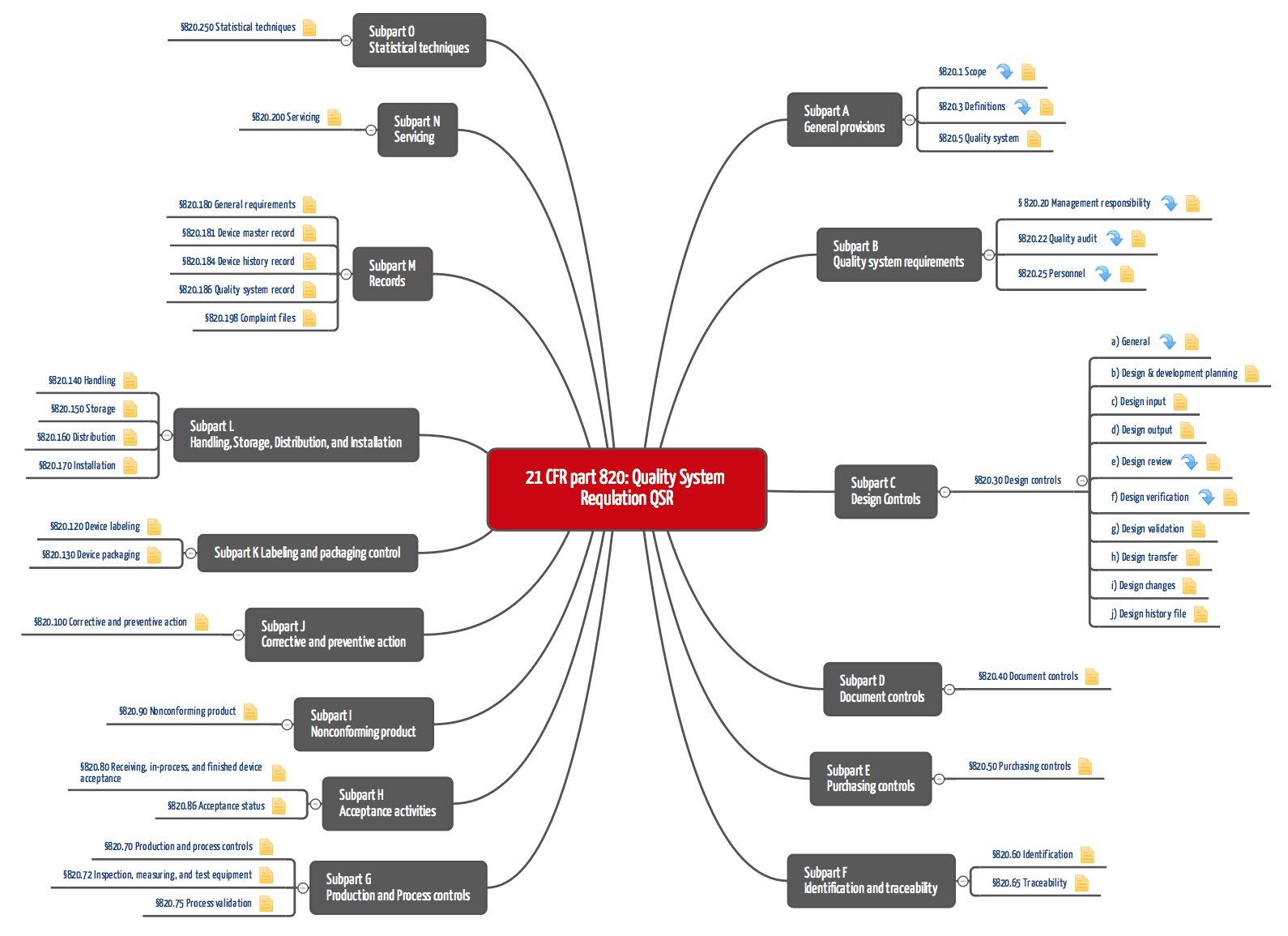
21 CFR Part 820 Subpart C – Design Controls - LearnGxP: Accredited Online Life Science Training Courses