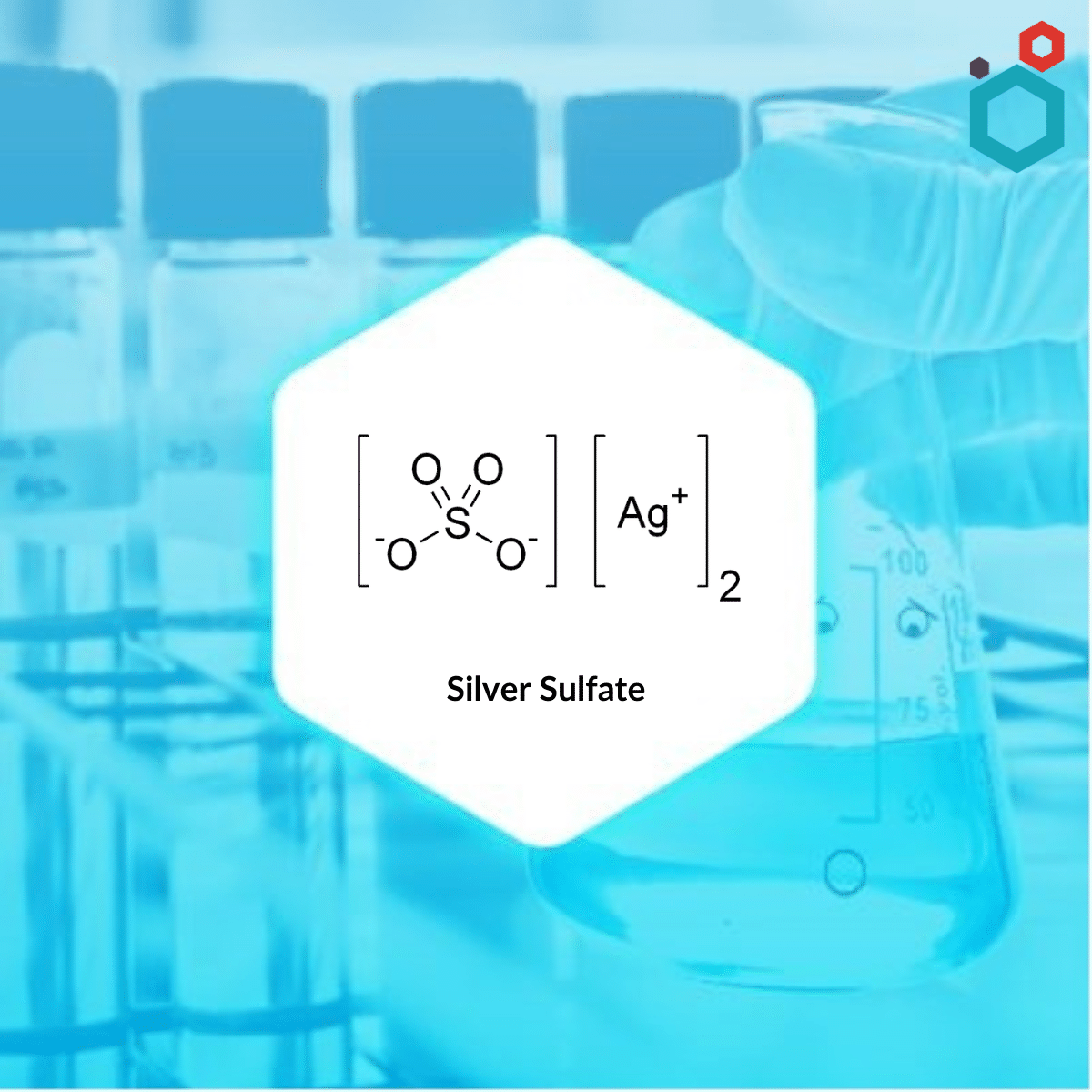
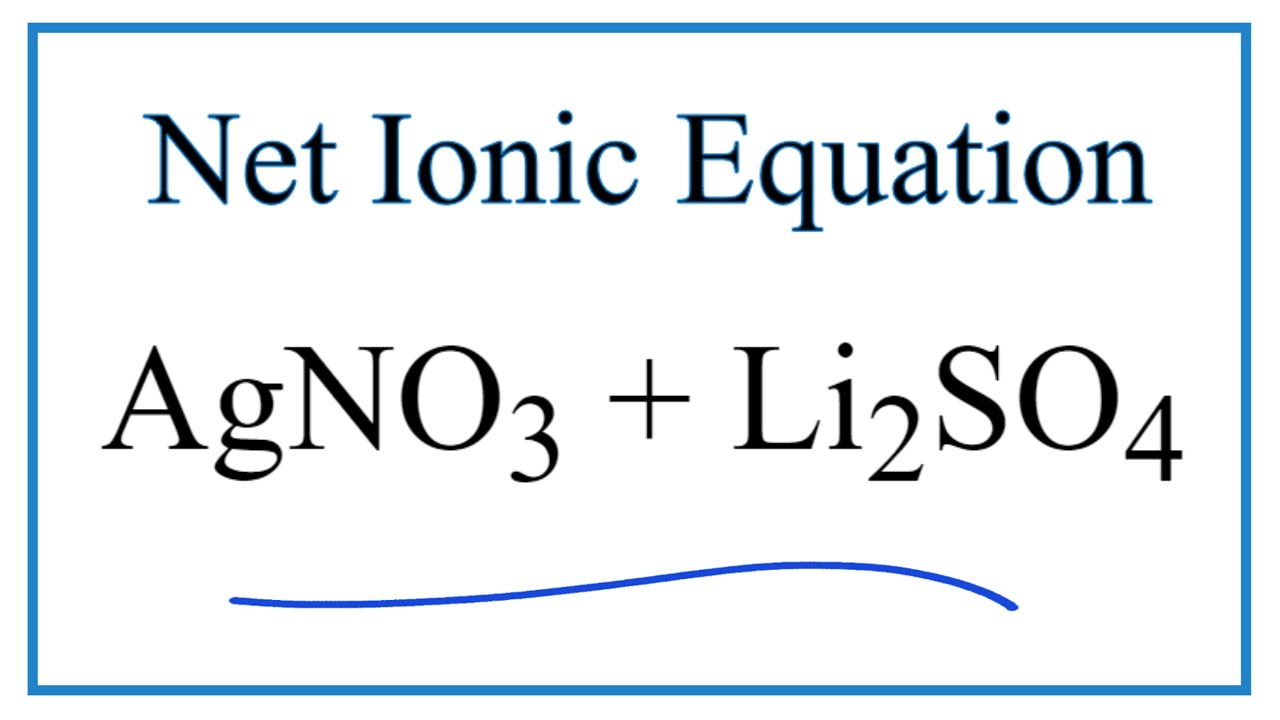Solubility Allows us to flavor foods -- salt & sugar. Solubility of tooth enamel in acids. Allows use of toxic barium sulfate for intestinal x-rays. - ppt download

Question Video: Identifying an Unknown Solution from the Reaction with a Sodium Sulfite Solution | Nagwa

Why does the color of Silver sulphate NPs change from yellow to dark violet without change to red color? | ResearchGate

Ag@Ag2SO4 nanoparticles: simple microwave-assistance synthesis, characterization and its co-photocatalytic degradation of methylene blue | Journal of Materials Science: Materials in Electronics

SOLVED: Precipitation of Ag2SO4 Chemical equation: 2 Ag(aq, 0.025 M) + SO4 (aq, 0.025 M) â†' 3 Ag2SO4(s) ΔG° = -28.094 kJ/mol ΔG°mn,298 K = ΔG°TIn,298 K + 2.303 RT log Q
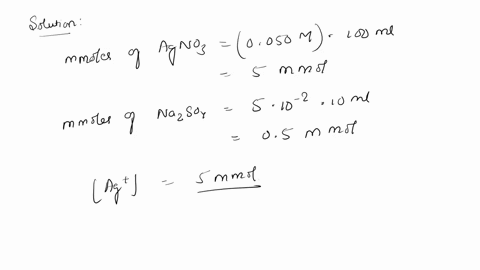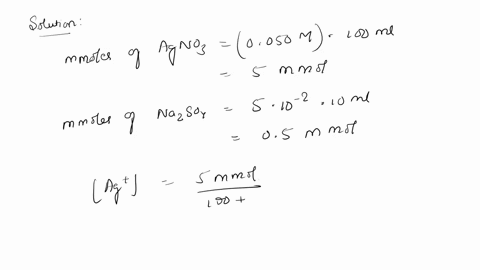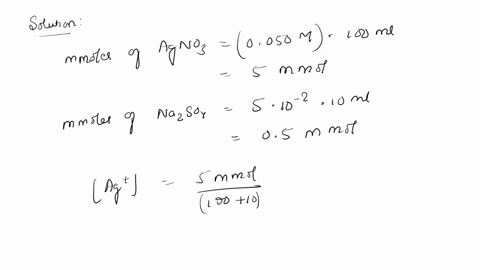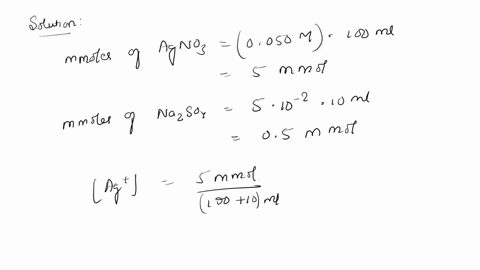
SOLVED: 10. Will silver (I) sulfate (Ag2SO4) precipitate when 100 mL of 0.050 M AgNO3 is mixed with 10.0 mL of 0.050 M Na2SO4? The solubility-product constant (Ksp) for Ag2SO4 is 1.5 * 10^-5.
%20at%20room%20temperature%20Iron%20(Fe)%20reacts%20with%20the%20acid%20producing%20hydrogen%20bubbles-%20Fe%20%2B%20H2SO4%20--%20.jpg)
Bildagentur | mauritius images | Iron reacts with sulfuric acid. Iron nail is placed in a test tube with 5M solution of sulfuric acid (H2SO4) at room temperature. Iron (Fe) reacts with

In the following reaction sequence in aqueous solution, the species X, Y and Z, respectively, are S2032- Ag Ag with time 9 X - Y Clear white black solution precipitate precipitate (A) (
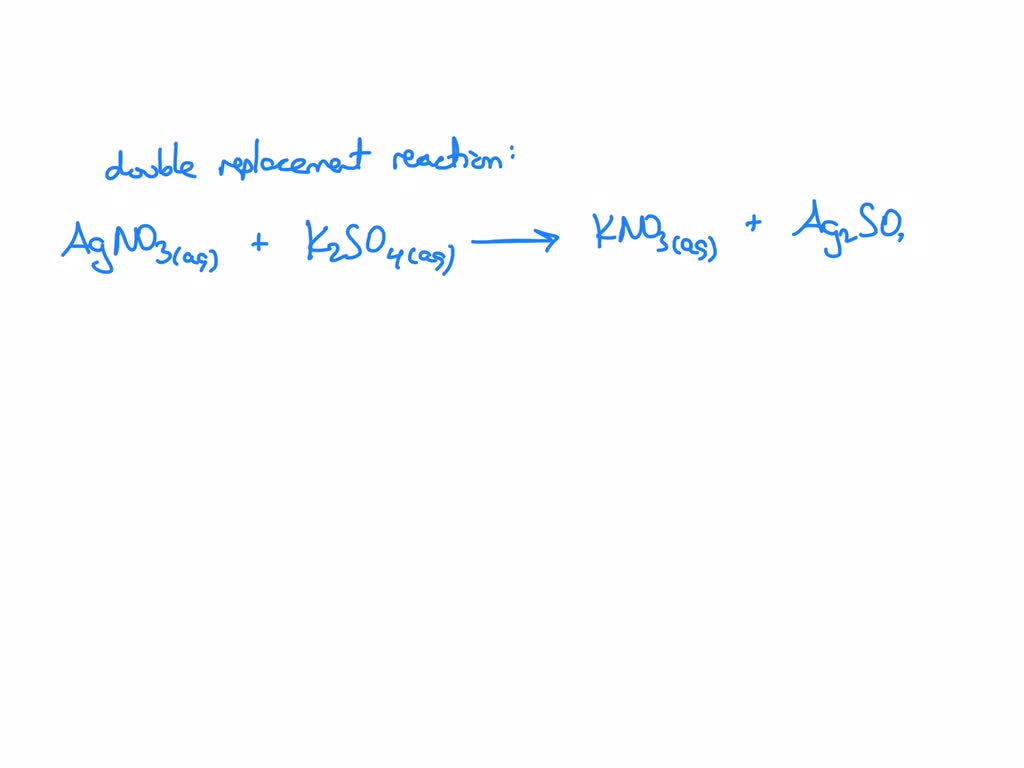
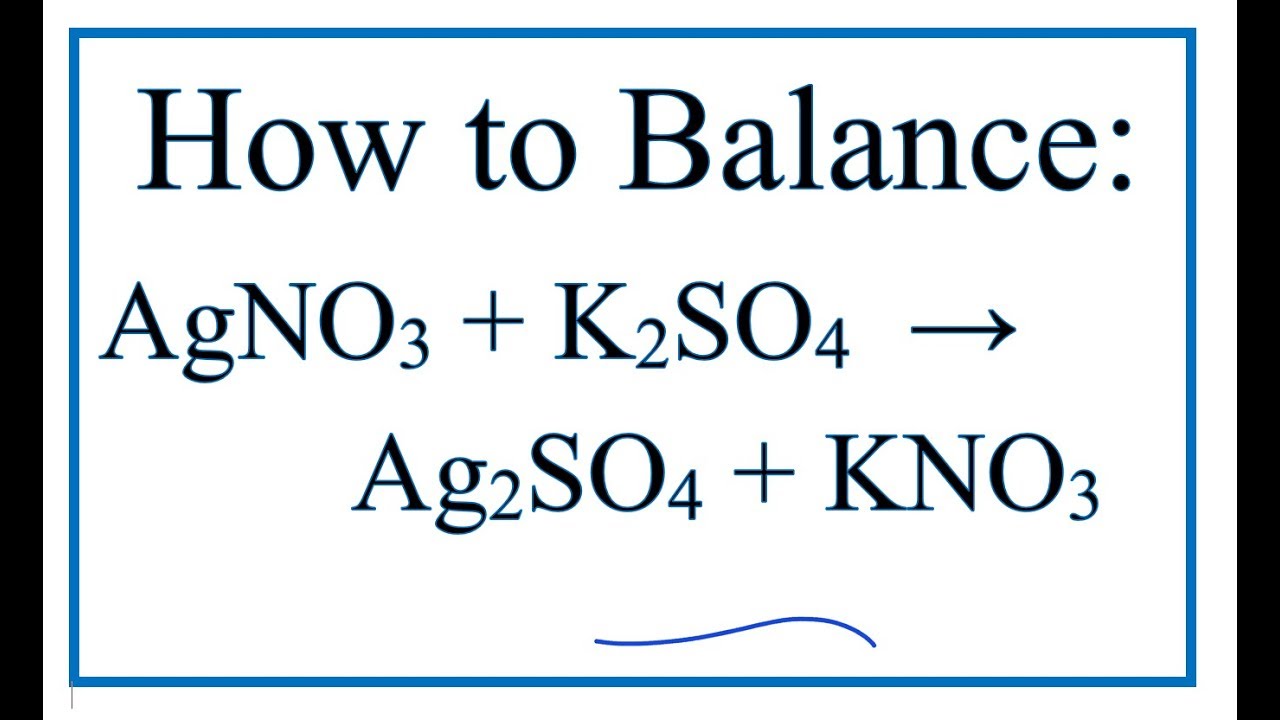
SOLVED: What occurs when aqueous silver nitrate, AgNO3, reacts with aqueous potassium sulfate, K2SO4?' Select one: Ag2SO4 forms as a precipitate. KNO3 forms as a precipitate. K2SO4 forms as a precipitate. No


![Ultimate Toolkit for HSC Chemistry Module 8 [Cheatsheet] | Learnable Ultimate Toolkit for HSC Chemistry Module 8 [Cheatsheet] | Learnable](https://www.learnable.education/wp-content/uploads/2020/05/Solubility-Rules.png)